 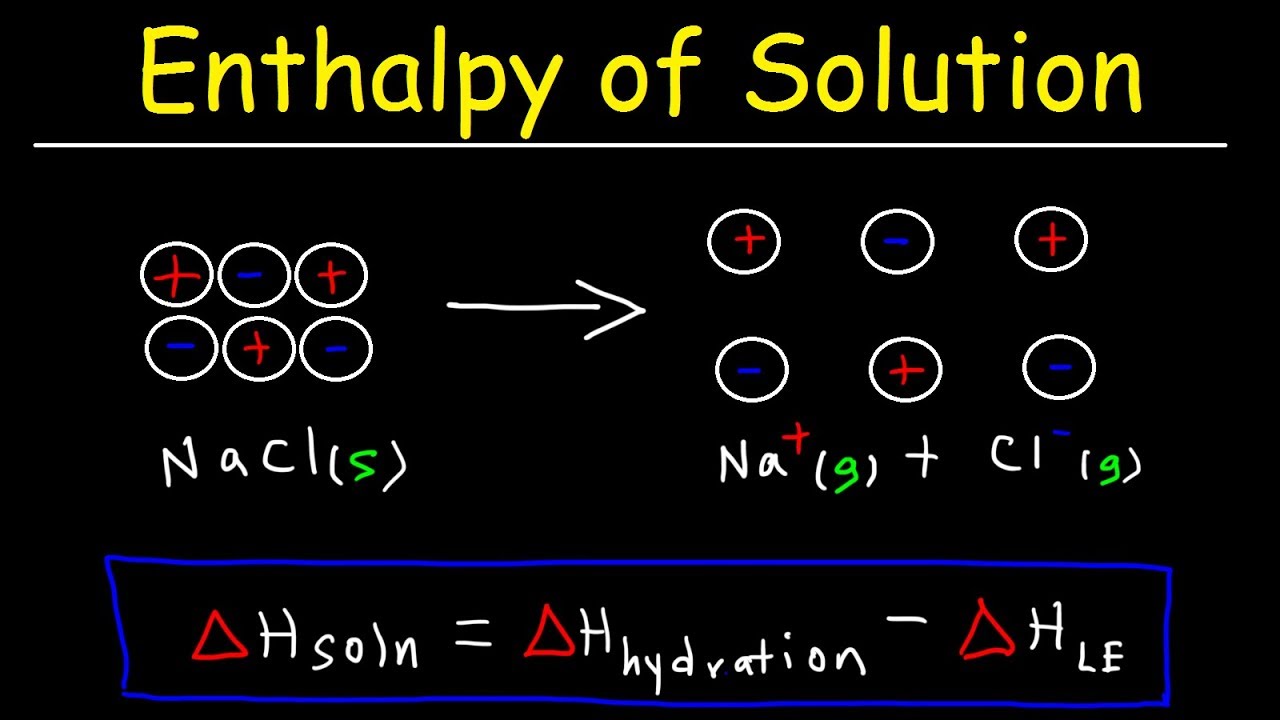
The explanation is based on the definition of lattice energy. Your question seems to be missing crucial information but we still tried to answer it as best as we could. The above solution had certain errors, please find the corrected solution below. The value of the lattice energy of the constituents of PbBr₂ (ionic solid) is not given here to explain the answer by showing calculations.Īs we said, we’re still missing the above information, so it might be good for you to resubmit your question and we’d be happy to try again! Thereby, the energy in the above reaction released in the formation of 1 mol of PbBr₂ (ionic solid) from its gaseous ions is the lattice energy. (ii) Formation of PbBr₂ (s) from its gaseous ions 
So, the energy in the above reaction used in converting 1 mol of PbBr₂ (ionic solid) into its gaseous ions is the lattice energy. (i) Separation of PbBr₂ into its gaseous ions The chemical equation representing the lattice energy for PbBr₂ (s) is as follows. Also, the energy released during the formation of one mole of an ionic solid from its constituent gaseous ions is also termed lattice energy. It is known that lattice energy is the amount of energy needed for the conversion of one mole of an ionic solid into its gaseous ionic components. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
